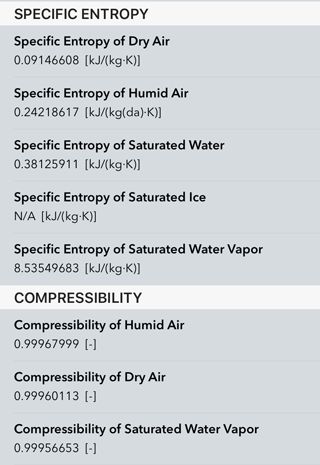
ACP - Reappraising the appropriate calculation of a common meteorological quantity: potential temperature
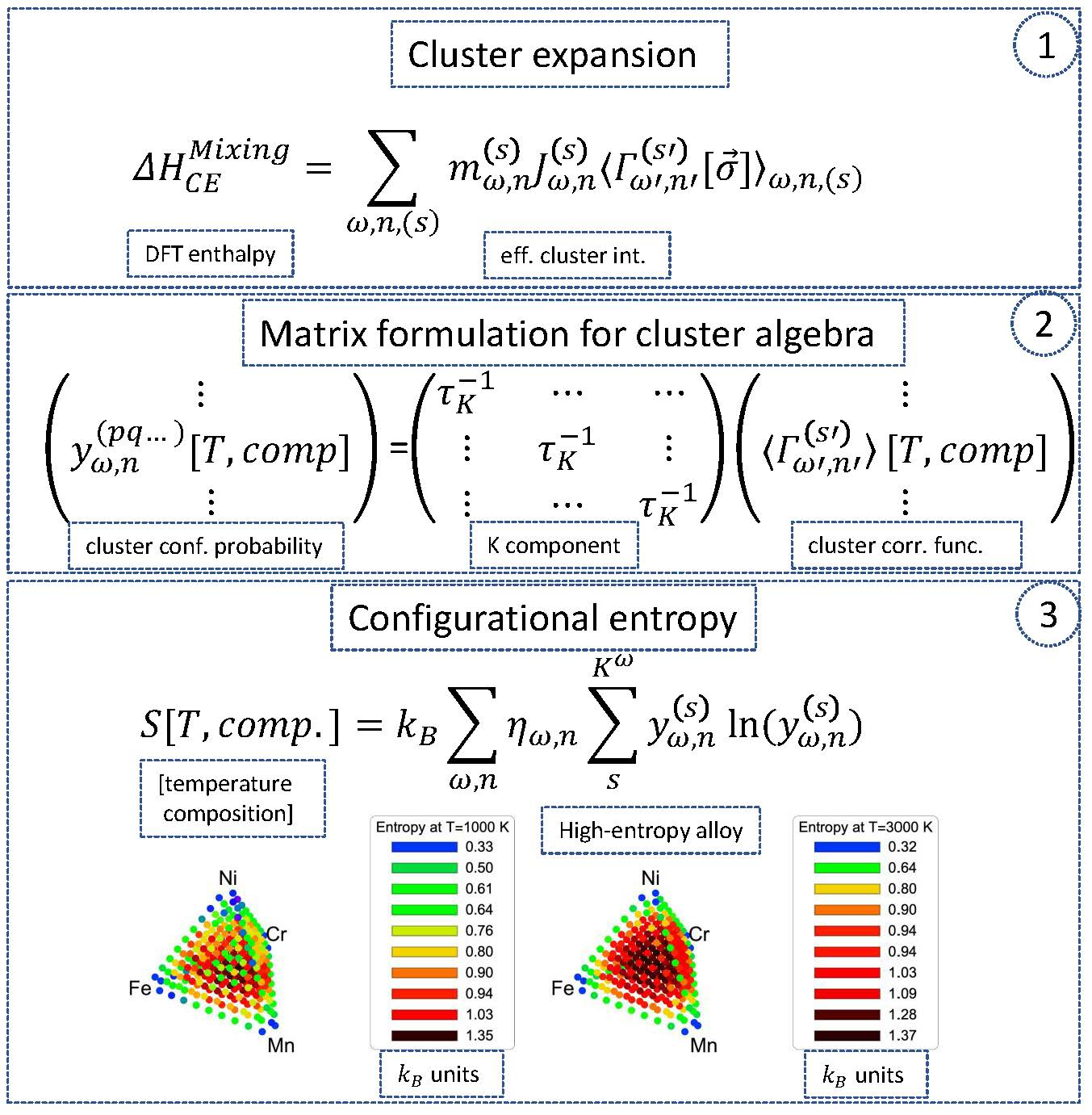
Entropy | Free Full-Text | Configurational Entropy in Multicomponent Alloys: Matrix Formulation from Ab Initio Based Hamiltonian and Application to the FCC Cr-Fe-Mn-Ni System

Calculate the increase in its Entropy for a liter of air if heated to double its volume P 3-28 - YouTube




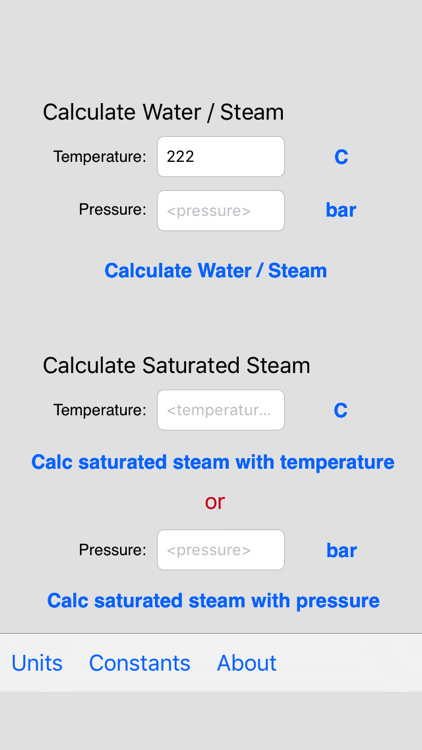


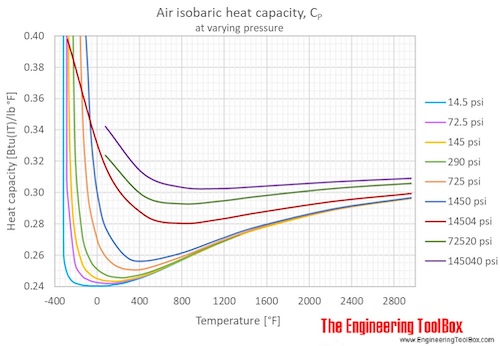


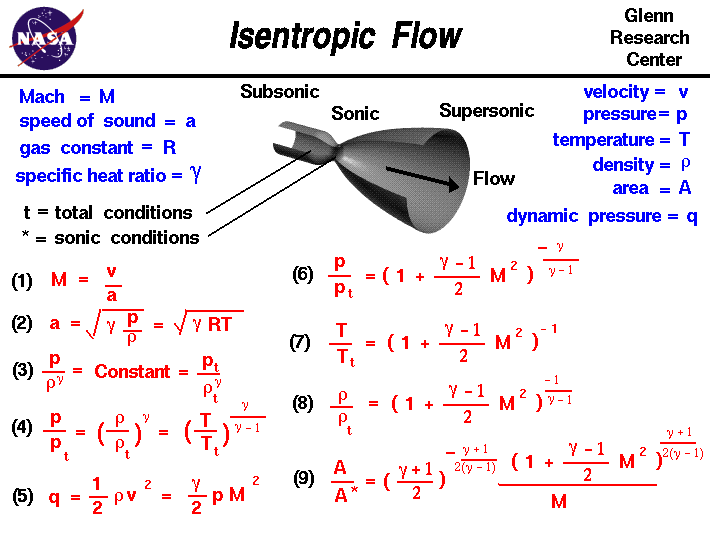
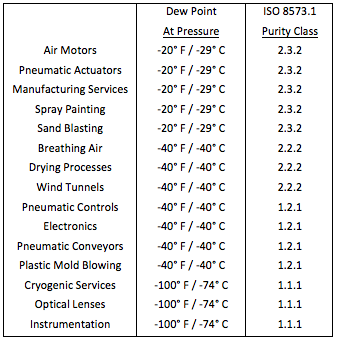
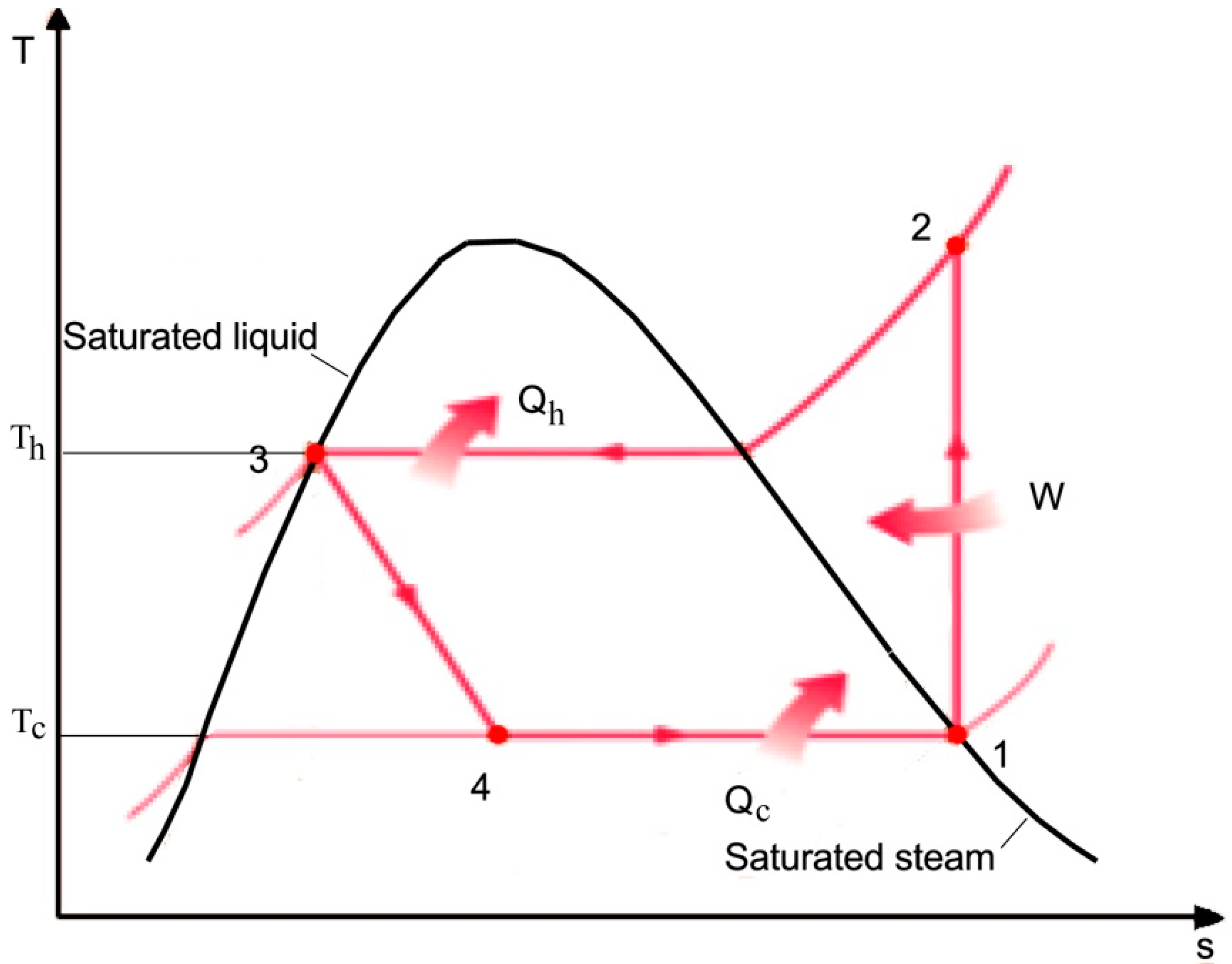
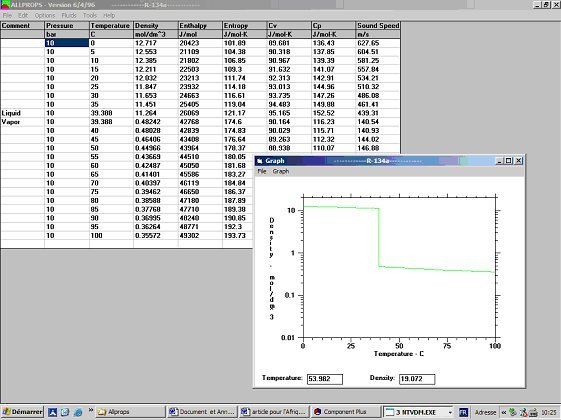
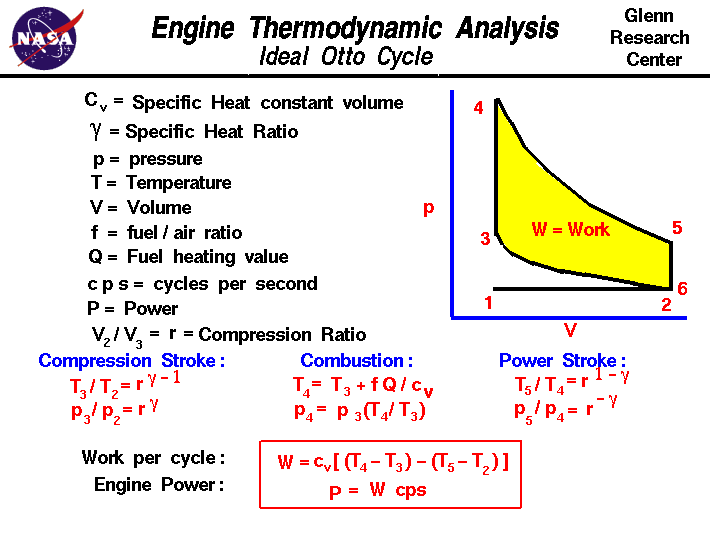

![PDF] Spreadsheet Calculations of Thermodynamic Properties | Semantic Scholar PDF] Spreadsheet Calculations of Thermodynamic Properties | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/6af9e71616fdfe0c904c310c6eb020d9e6eb417d/3-Figure2-1.png)