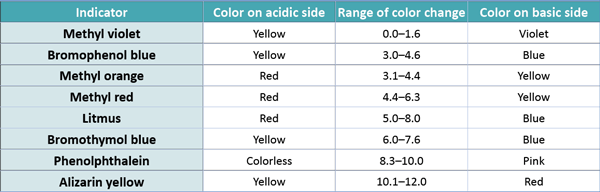
SOLVED: A cyanide ion-selective electrode obeys te equation E= constant 0.059 log/CN-| The electrode potential was -0.220 V when the electrode was immersed in 0.0010 M NaCN Estimate the constant in the

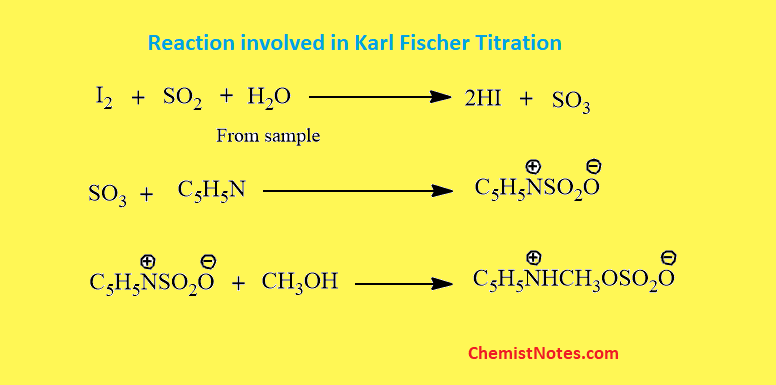
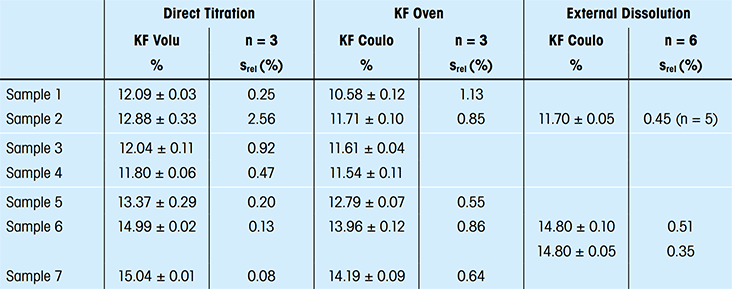
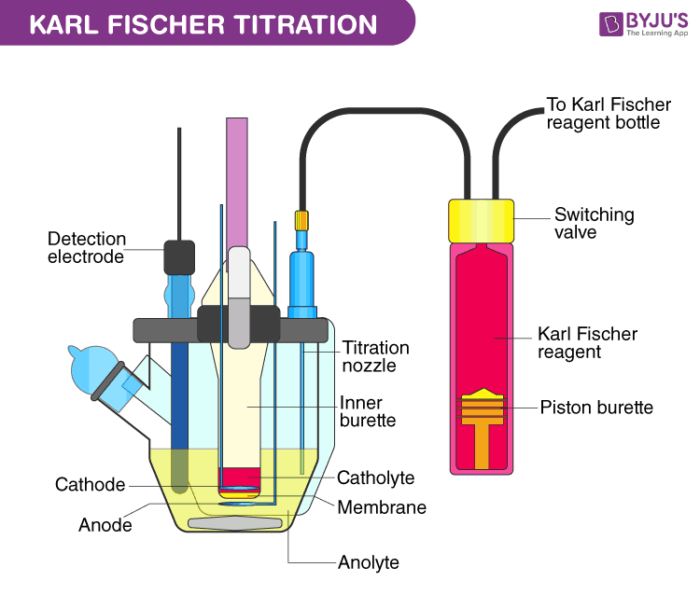
Pharmaceuticals Validation and stability-Technical Quality and Compliance: Water determination by Karlfischer reagent(KF)







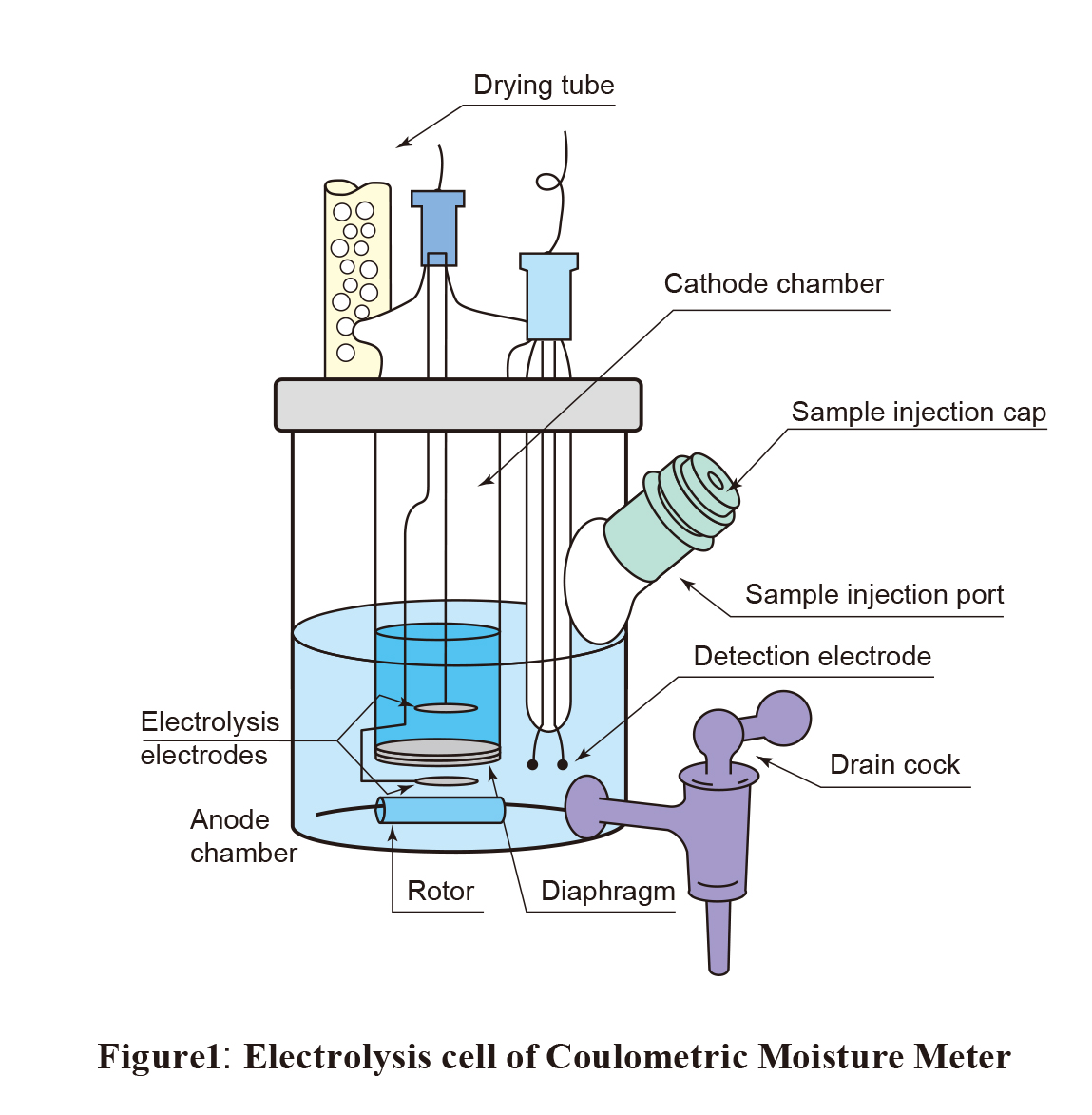

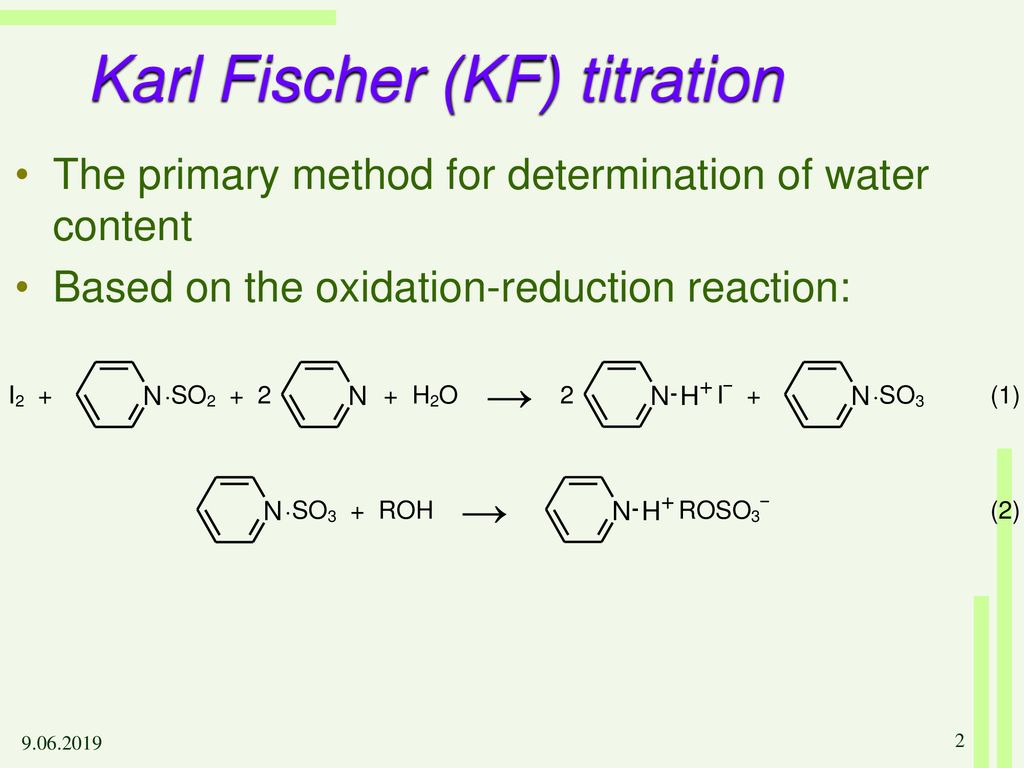


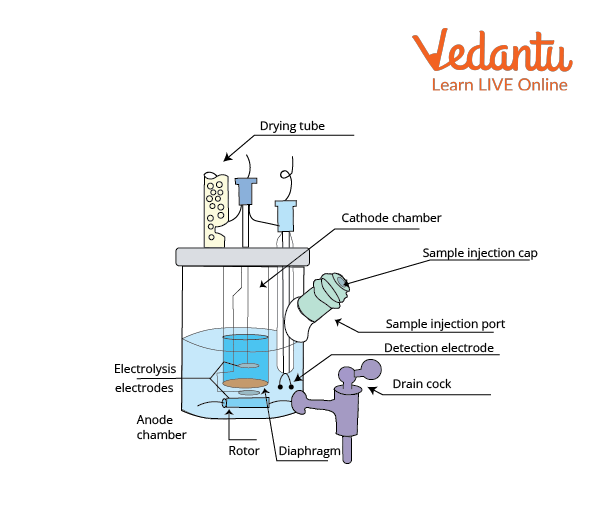

.gif)