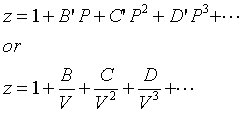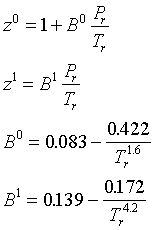Calculate the Pressure Using Viral Equations in Physical Chemistry | Chem 331 | Assignments Fisicoquímica | Docsity
![Virial equation is: PV(M)=RT[A+(B)/(V(M))+(C )/(V(M^(2)))+…], where A, B, C, …. are first second,third, … virial coefficent, respectively, For an ideal gas Virial equation is: PV(M)=RT[A+(B)/(V(M))+(C )/(V(M^(2)))+…], where A, B, C, …. are first second,third, … virial coefficent, respectively, For an ideal gas](https://d10lpgp6xz60nq.cloudfront.net/ss/web/298832.jpg)
Virial equation is: PV(M)=RT[A+(B)/(V(M))+(C )/(V(M^(2)))+…], where A, B, C, …. are first second,third, … virial coefficent, respectively, For an ideal gas

SOLVED: 1.For a obeying the equation of state p RT gas evaluate J2 and Or , Vm b Vm RT 1 + B 2. The virial equation of state is: pVm Vm +

Deriving Second Osmotic Virial Coefficients from Equations of State and from Experiment | The Journal of Physical Chemistry B

PPT - Advanced Thermodynamics Note 2 Volumetric Properties of Pure Fluids PowerPoint Presentation - ID:756864